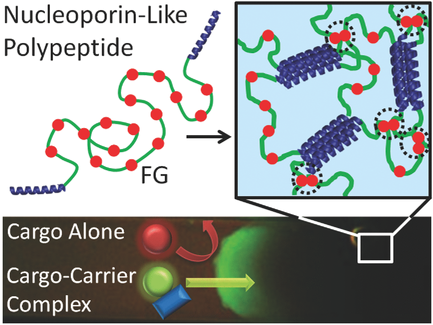
The development of polymer gels that are able to control transport of cargo is an exciting platform technology for a range of applications including protein purification, blood diagnostics and sensing, and toxin removal from biological systems. The nuclear pore complex, which spans the nuclear envelope and enables selective transport of nuclear transport receptor (NTR) proteins and their cargo, contains phenylalanine-glycine-rich (FG) nucleoporin proteins. We have developed nucleoporin-inspired proteins based on a composite repeat sequence of the yeast nucleoporin that were flanked with associative pentameric coiled-coil domains (P) to form a hydrogel. These new gels demonstrated rapid diffusion of proteins fused with NTR proteins and slower diffusion of non-NTR-fused proteins. Furthermore, a molecular transport theory was shown to account for the selective transport with a walking mechanism. Moving forward, we are probing consensus repeat sequences of non-yeast nucleoporins to enable the selective transport of additional cargo.
Highlighted Publications
1. Yang, Y. J., Mai, D. J., Dursch, T. J., & Olsen, B. D. (2018). Nucleopore-inspired polymer hydrogels for selective biomolecular transport. Biomacromolecules, 19(10), 3905-3916. Link
2. Kim, M., Chen, W. G., Kang, J. W., Glassman, M. J., Ribbeck, K., & Olsen, B. D. (2015). Artificially engineered protein hydrogels adapted from the nucleoporin Nsp1 for selective biomolecular transport. Advanced Materials, 27(28), 4207-4212. Link